ネイルチップ ツイードネイル 秋冬 キルティングネイル ガーリー
(税込) 送料込み
商品の説明
商品説明
ネイルチップ3850円♥ご購入前にショップ情報をお読みください
♥こちらはオーダー専用です♡
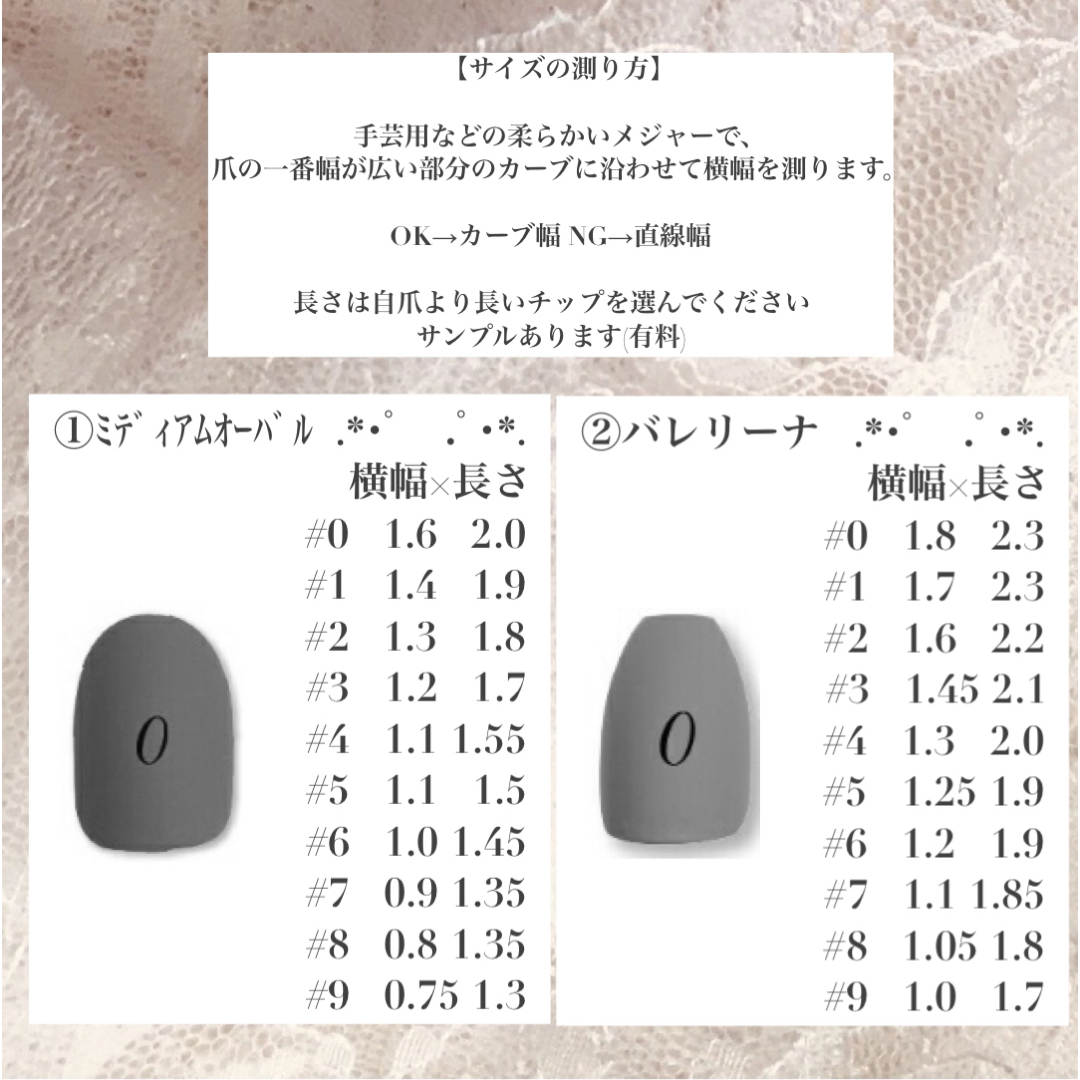
チップの種類とサイズコメントにてご指定ください。
専用ページご用意させていただきます
現品はSOLDOUTです
♥定形外郵便送料込
入金確認後7~14日前後で発送します
→追跡無し、土日祝は配送ありません
→オプションでゆうパケット又は速達可能です
⋱⋰⋱⋰⋱⋰⋱⋰⋱⋰⋱⋰⋱⋰⋱⋰⋱⋰⋱⋰⋱⋰
・ゆうパケットポスト(入金確認後2日以内に発送)【+250円】
・現品ネイル速達(入金確認後2日以内発送)
【+500円】
・お急ぎ作成速達(再作成のみ、1週間以内に発送)【+1200円】
・超特急作成速達(再作成のみ、3日以内に発送)【+1500円】
・両面テープ3回分【+200円】
・両面テープ5回分【+300円】
・トップ二重コート【+200円】
・イニシャル追加筆記体(白、黒、ゴールド)
【1文字+50円】
・イニシャル追加3Dひらがな筆記体(色指定可能)
【1文字+200円】
・色変更、デザイン変更、パーツ追加(1箇所)
【+200円~】
・おまとめ割(オーダー・現品ネイル対象)
2セット→50円引き
3セット→100円引き
・インスタフォロー割mirua.nail
(購入前に申告お願いいたします)
100円引き
⋱⋰⋱⋰⋱⋰⋱⋰⋱⋰⋱⋰⋱⋰⋱⋰⋱⋰⋱⋰⋱⋰⋱
#ネイルチップ
#ネイル
#量産型ネイル
#韓国ネイル
#ワンホンネイル
#量産型ネイルチップ
#ガーリーネイル
#ツイードネイル
#リボンネイル
#キルティングネイル
#秋ネイル
#埋めつくしネイル
mirua.nail
39999円ネイルチップ ツイードネイル 秋冬 キルティングネイル ガーリーコスメ/美容ネイル2024年春】ガーリー ツイードのネイルデザイン集|人気順韓国ネイルネイルチップ ツイードネイル ガーリーネイル 秋冬ネイル
韓国ネイルネイルチップ ツイードネイル ガーリーネイル 秋冬ネイル
つけ爪/ネイルチップネイルチップ ツイードネイル ガーリーネイル 秋冬
2024年春】ガーリー ツイードのネイルデザイン集|人気順
2024年春】ガーリー ツイードのネイルデザイン集|人気順
ネイルチップ ツイードネイル 秋冬 キルティングネイル ガーリー韓国
No.30 ぷっくりキルティング🎀🖤ツイード♡ ネックレス モチーフ
秋/冬/女子会/ハンド/ツイード - o.risatoのネイルデザイン[No.8382017
ネイルチップ ツイードネイル 秋冬 キルティングネイル ガーリー - ネイル
ネイルチップ ツイードネイル 秋冬 キルティングネイル ガーリー - ネイル
韓国ネイルネイルチップ ツイードネイル ガーリーネイル 秋冬ネイル
2024年春】ツイード ロングのネイルデザイン集|人気順
冬/クリスマス/ハンド/ツイード/キルティング - o.risatoのネイル
2024年春】キルティングのネイルデザイン集|人気順|ホットペッパー
ネイルチップ ツイードネイル ガーリーネイル 秋冬ネイル-
秋/冬/ハンド/ツイード/キルティング - o.risatoのネイルデザイン[No
ネイルチップ ツイードネイル 秋冬 キルティングネイル ガーリー - ネイル
ぷっくりキルティングネイルが今の気分。カラバリ豊富なトレンド
冬/ハンド/ツイード/キルティング/アイシング - 奈奈のネイルデザイン
ネイルチップ ツイードネイル ガーリーネイル 秋冬ネイル-
ネイルチップ ツイードネイル 秋冬 キルティングネイル ガーリー - ネイル
キルティングネイルのネイルデザイン(46ページ目)|ネイルブック
お値下げ中 秋冬ネイル ツイードネイル ブラック 量産型 ガーリー
ネイルチップ ツイードネイル 秋冬 キルティングネイル ガーリー - ネイル
キルティングネイルがぷっくりかわいい♡冬におすすめなデザイン
2024年春】ガーリー 冬のネイルデザイン集|人気順|ホットペッパー
ネイルチップ ツイードネイル ガーリーネイル 秋冬ネイル-
No.64】水玉♡ガーリー リボン ネイルチップ 量産型 地雷系の通販 by
秋/冬/クリスマス/女子会/ハンド - angenail_MAKIのネイルデザイン[No
韓国ネイルネイルチップ ツイードネイル ガーリーネイル 秋冬ネイル
2024年春】ガーリー ツイードのネイルデザイン集|人気順
フレンチガーリーネイル 黒フレンチネイル キルティング - www
お値下げ中 秋冬ネイル ツイードネイル ブラック 量産型 ガーリー
即納可 現品ネイルチップ ツイードネイル 秋冬 キルティングネイル
No.64】水玉♡ガーリー リボン ネイルチップ 量産型 地雷系の通販 by
ツイード/キルティング/モノトーンのネイルデザイン|ネイルブック
韓国ネイルネイルチップ ツイードネイル ガーリーネイル 秋冬ネイル
布感ツイード❤️ ・ ・ #ツイードネイル#布感ツイード#赤ネイル
キルティングツイードネイル | Ethical B. Tokyo(エシカルビー
秋冬の定番といえば!ツイード柄ネイル|2023秋ネイル
商品の情報
メルカリ安心への取り組み
お金は事務局に支払われ、評価後に振り込まれます
出品者
スピード発送
この出品者は平均24時間以内に発送しています